|
This is the reason all orbitals can hold a maximum of two electrons. Therefore, if two electrons occupy the same orbital, they must have different spins. Rule 3 (Pauli-Exclusion Principle): Each electron is described with a unique set of four quantum numbers (a unique address).For example, all three p orbitals in a p subshell will have one electron before a single p orbital contains two electrons. Furthermore, the most stable configuration results when the spins are parallel (i.e. same shell and subshell), they must first singly occupy (half-fill) each empty orbital in a subshell before double occupying (completely filling) them. Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula NaOH.It is a white solid ionic compound consisting of sodium cations Na + and hydroxide anions OH. Rule 2 (Hund's Rule): When electrons occupy degenerate orbitals (i.e.Sodium is not found free in nature, but sodium compounds are common. Within these shells, an s subshell is the lowest energy followed by p, then d, then f. Sodium is the 6th most abundant element in the Earths crust, making up approximately 2.6 of the earth, air, and oceans. Shells increase in energy in order from 1 to 2 to 3, and so on. Rule 1 (Aufbau Principle): Electrons occupy the lowest-energy orbitals (closest to the nucleus) possible, starting with 1 s, then 2 s, 2 p, and continuing on to higher energy (further away from the nucleus).There are a set of general rules that are used to figure out the electron configuration of an atomic species: Aufbau Principle, Hund's Rule and the Pauli-Exclusion Principle. (CC BY-SA 2.0 UK Greg Robson modified by Pumbaa via Wikipedia) The sodium atom donates its valence electron to the chlorine atom and the chlorine atom receives that electron.\): Shell diagrams of hydrogen (H), helium (He), lithium (Li), and Berryellium (Be) atoms. The electron configuration of chlorine shows that the valence electrons of chlorine are seven. Sodium with the Rare Earth Elements, Cluster-type Lithium Lanthanide Double. This valence electron participates in the formation of bonds with atoms of other elements. To write the orbital diagram for the Sodium atom (Na) first we need to write the electron configuration for just Na. diagram and spectroscopic studies of mixed oxides of lanthanides and. We know that the valence electron in sodium is one. Sodium participates in the formation of bonds through its valence electron. Since the last shell of a sodium-ion has eight electrons, the valence electrons of sodium ion(Na +) are eight. That is, in this case, the valency of the sodium ion is +1. The electron configuration shows that the sodium atom has acquired the electron configuration of neon. The electron configuration shows that sodium ion(Na +) have only two shells and the last shell has eight electrons. 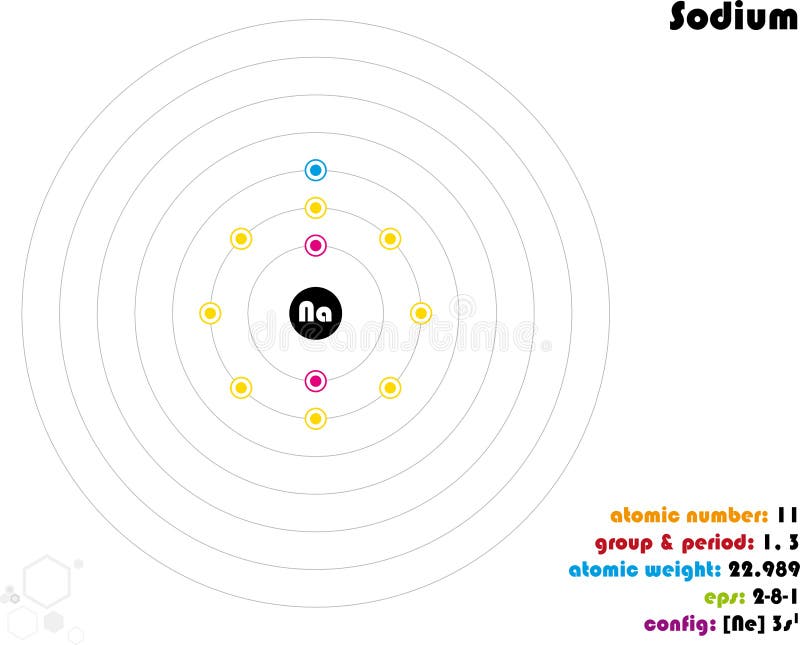
The electron configuration of sodium ion(Na +) is 1s 2 2s 2 2p 6. 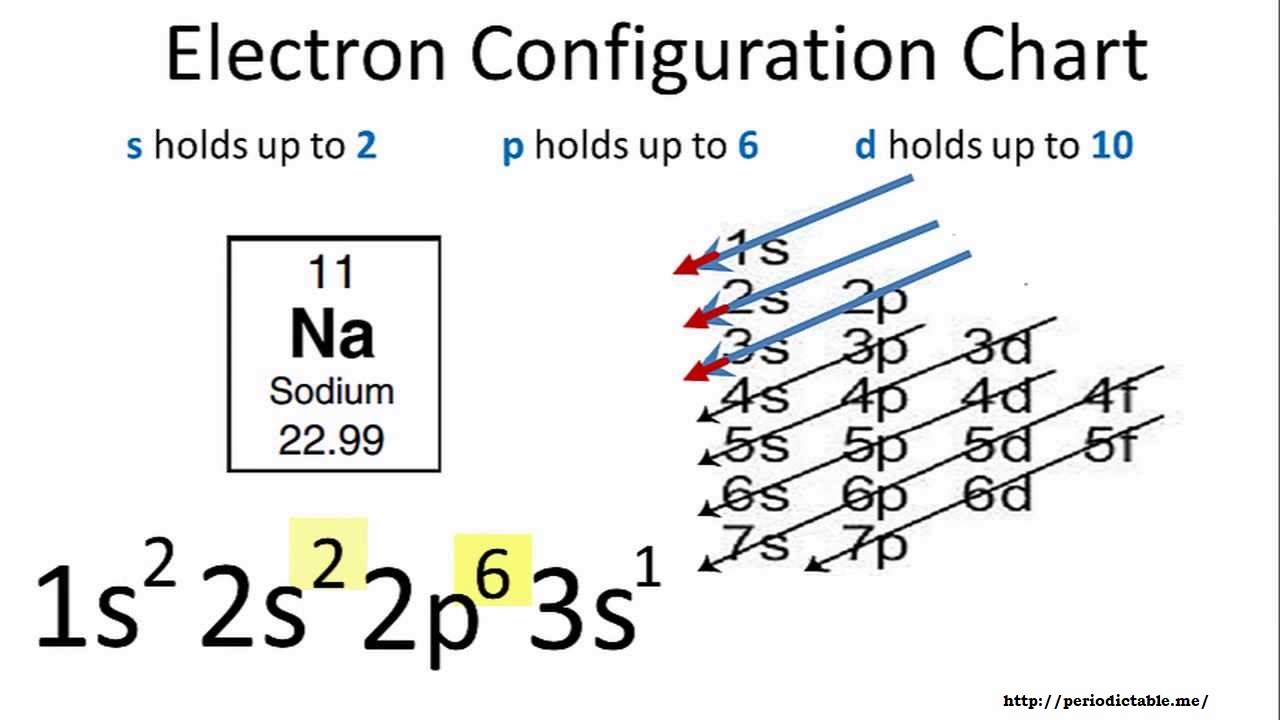
Sodium donates the electron of the last shell to form bonds and turns into a sodium ion(Na +). Magnesium is neutral and its atomic number is 12, hence, the number of protons and electrons available for its Bohr diagram is also 12. Draw a diagram of a plant cell and label at least eight class. These dots are arranged to the right and left and above and below the symbol. Note: The number of electrons in sodium atom and sodium ion is 11 and 10 respectively. The number of dots equals the number of valence electrons in the atom. The elements that form bonds by donating electrons are called cations. The Bohr model of Magnesium (Mg) is drawn with three electron shells, the first shell contains 2 electrons, the second shell contains 8 electrons and the third shell contains 2 electrons. A Lewis electron dot symbol (or electron dot diagram or a Lewis diagram or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The elements that have 1, 2, or 3 electrons in the last shell donate the electrons in the last shell during bond formation. How many valence electrons does sodium ion(Na +) have? The electron configuration of sodium shows that there is an unpaired electron in the last orbit of sodium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
